Retinal diseases are the major cause of blindness in industrialized countries. For example, an estimated total of 196 million people will be affected by age related macular degeneration in 2020. While tremendous effort is being made to develop novel therapeutic strategies to rescue retinal neurons and the retinal pigment epithelium, optimal means for evaluating the effects of such treatments are still missing.
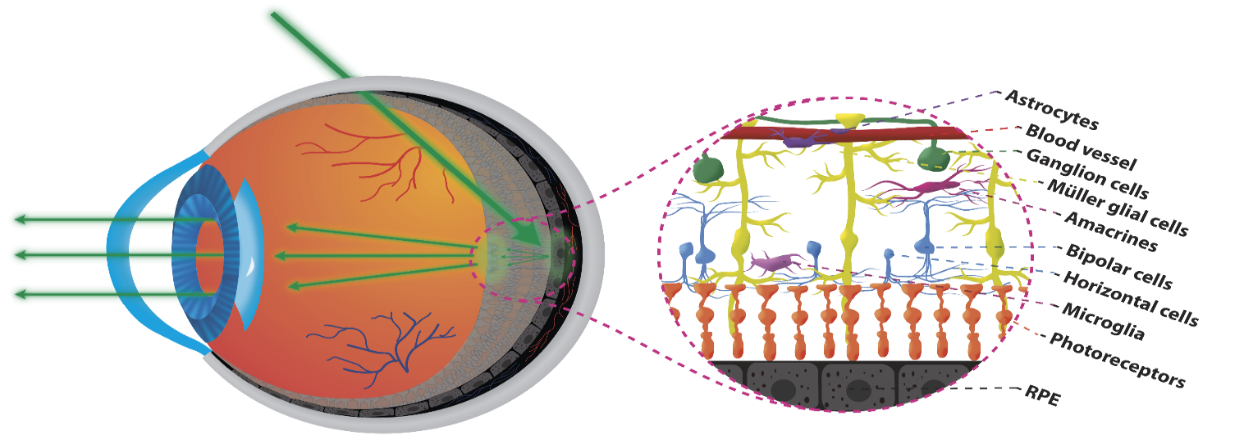
Transscleral oblique illumination (left) – Representation of the cellular structures of the human retina (right).
The evaluation and monitoring of cells health in the human retina is crucial for following the time course of retinal diseases, detecting lesions before irreversible visual loss occurs, and evaluating the effects of treatments. Despite the phenomenal advances in optical coherence tomography (OCT) and adaptive optics systems, in vivo imaging of several retinal cells is limited by the fact that the cell contrast achieved via reflection is extremely low.

In-vivo human RPE cells acquired with TOPI.

Transscleral illumination as performed in TOPI.
We developed a radically different method compared with OCT to visualize retinal cells with high contrast, high resolution, and within an acquisition time suitable for clinical use thanks to a new technique called transscleral optical phase imaging (TOPI). TOPI uses high-angle oblique illumination of the retina to enhance the contrast of the transparent cells and of the retinal pigment epithelium. We demonstrated the feasibility of the proposed imaging modality on ex vivo retinas of humans, rats and pigs and applied it in vivo on humans (approval from the ethical commission from Canton de Vaud to perform a proof of concept study on human subjects, CER-VD N°2017-00976), without pupil dilation by combining TOPI with adaptive optics. Our results show the ability of TOPI to image and quantify retinal cells up to the retinal pigment epithelium depth within a maximum time of 19 seconds on 7 human subjects.
Collaborators:
Prof. Francine Behar-Cohen (Inserm, U1138, Team 17, From physiopathology of ocular diseases to clinical development, Paris Descartes University, Sorbonne Paris Cité, Centre de Recherche des Cordeliers, and Ophtalmopole, Hôpital Cochin, Assistance Publique Hôpitaux de Paris, Université Paris Descartes, Paris, France).
Dr. Laura Kowalczuk (Faculty of Biology and Medicine, University of Lausanne, and Jules-Gonin Eye Hospital, Fondation Asile des aveugles, Lausanne, Switzerland).
Funding : EPFL Innogrant, Gebert Ruef Stiftug, EIT Health.
- https://www.earlysight.com/
- https://www.grstiftung.ch/en/media/portfolio~grs-052-17~.html
- https://eithealth.eu/project/retinal-phase-contrast-imaging-for-early-diagnosis/
Publications:
Contacts : Timothé Laforest, Mathieu Künzi, Christophe Moser
Support:
 |
 |
 |
 |
 |
 |