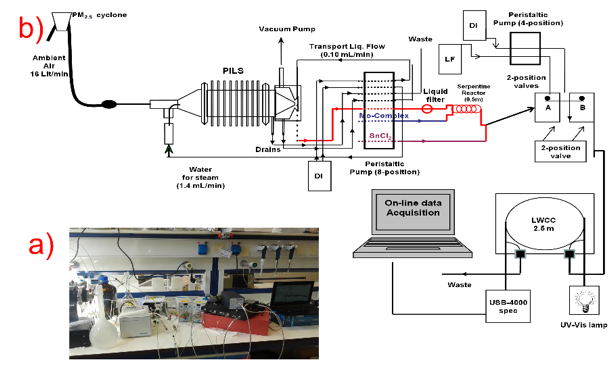
Primary productivity of continental and marine ecosystems is often limited or co-limited by phosphorus. The atmosphere is considered a principal source of externally-supplied nutrients for large areas of the surface ocean, and oligotrophic open oceans. Atmospheric inorganic P species (e.g. mono- or diprotonated orthophosphate) comprise the most bioavailable P form, and have been studied for many decades. Inorganic P are currently studied using filter samples with poor temporal resolution (1 sample/day or longer) and potentially significant sampling artifacts. To address the above limitation, we have developed an automated online system that measures P in atmospheric particles spectrophotometrically, using the molybdenum blue complex method (Violaki et al., 2016). The system consists of a Particle-In-Liquid sampler coupled with a reaction coil and a liquid capillary waveguide detection cell.
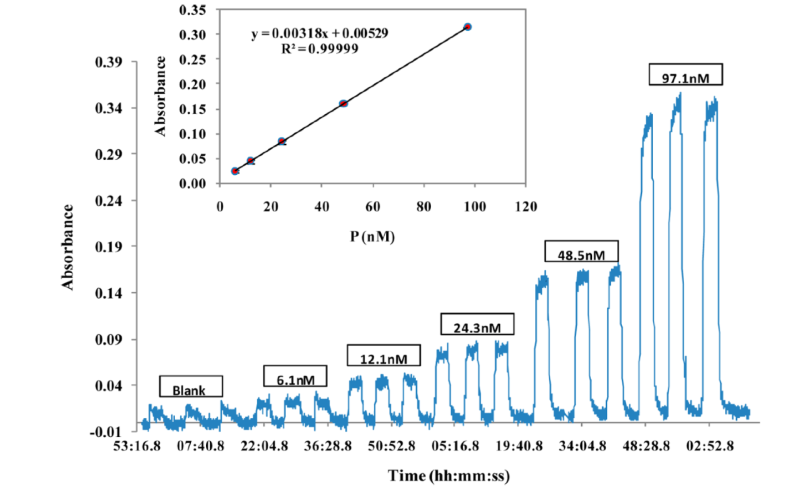
This novel technique offers at least an order of magnitude enhancement in sensitivity over existing approaches allowing for SRP measurements of unprecedented frequency (6-8 min), leading to greater understanding of the sources and impacts of SRP in atmospheric chemistry. The detection limit was 0.4 nM P, equivalent to 0.03 nmol P m−3 in atmospheric particles.