Light dosimetry and delivery
The performance of light-based therapies such as photodynamic therapy (PDT) and photobiomodulation (PBM) therapy depends, among other factors, on the accurate delivery and monitoring of light to the biological tissues. Thus, our activities aim at:
- Developing light delivery systems for specific applications. These light distributors are optimized to deliver homogenous and controlled light doses to the targeted tissues with minimal losses. Simplicity, compatibility with commercial light sources and medical devices, such as guiding catheters or modern endoscopes, as well as cost and safety are important parameters for their design. Some of these developments led to the foundation of one of our spin-off company, Medlight SA [http://www.medlight.com/].
- Developing measurement setups to characterize the radiometric properties of light distributors.
- Developing new instruments for the in situ measurement of the light dose and fluence rate in real-time.
- Measuring the optical parameters of biological tissues.
- Modeling the propagation of light in complex organs by Monte-Carlo simulations.
These developments and studies enable us to deliver light with accurate dosimetry in many situations.
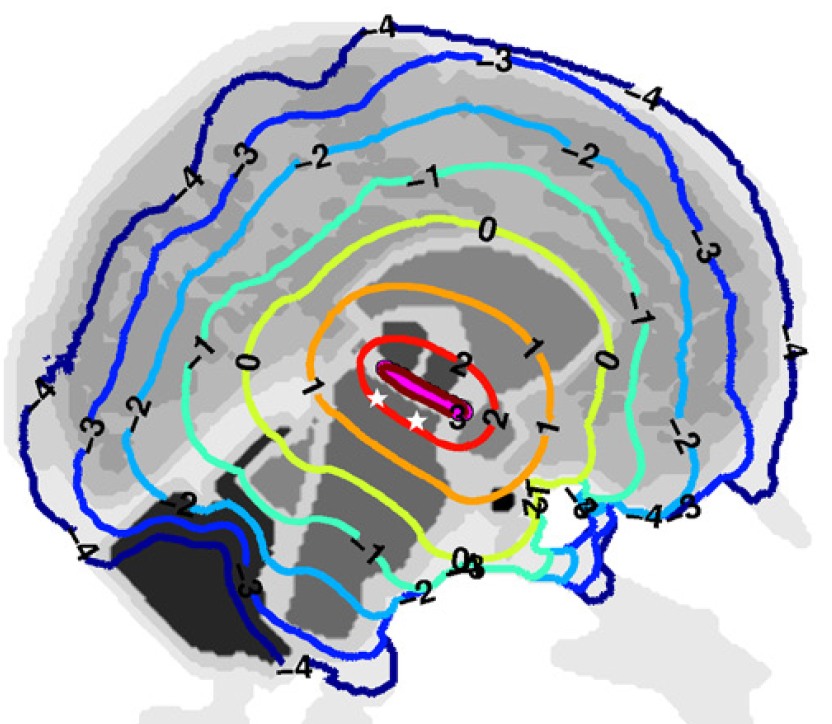 Light fluence rate distribution in a human brain when a cylindrical light diffuser (magenta line: 20 mm length and 1 mm diameter) is inserted in the third ventricle. Colored contour lines represent the log10 fluence rate computed from optical tissue coefficients at 808 nm derived from “Pitzschke et al., Phy. Med. Biol., 60, pp 2921-27, 2015” when the irradiance delivered by the light diffuser is 1 W/cm2. The white stars represent the location of the substantia nigra pars compacta.
Light fluence rate distribution in a human brain when a cylindrical light diffuser (magenta line: 20 mm length and 1 mm diameter) is inserted in the third ventricle. Colored contour lines represent the log10 fluence rate computed from optical tissue coefficients at 808 nm derived from “Pitzschke et al., Phy. Med. Biol., 60, pp 2921-27, 2015” when the irradiance delivered by the light diffuser is 1 W/cm2. The white stars represent the location of the substantia nigra pars compacta.
Photobiomodulation for the treatment of Parkinson’s disease
Numerous pre-clinical and clinical studies document the therapeutic effect of photobiomodulation (PBM) for the treatment of various conditions, including central nervous system disorders. In addition, there is a growing body of evidence showing that PBM modulates mitochondrial function by stimulating complex IV-related respiration and other metabolic processes.
Parkinson’s disease (PD) is a neurodegenerative disease characterized by abnormal mitochondrial function. Currently, available treatment options only alleviate the symptoms of PD.
Promising pre-clinical studies performed by our group and others show the neuroprotective and neuro(re)generative potential of PBM for the treatment of PD. In particular, in collaboration with Dr. Patrik Verstreken (VIB, Belgium), we have demonstrated that PBM restores locomotor function in PINK1 mutant flies. We have also shown that the effect is dose-dependent and is maximal between 5 and 24 hours after treatment. Using the same model, we have shown for the first time in vivo a significant increase in mitochondrial membrane potential and ATP production.
The goal of this project is to develop and optimize various concepts for the treatment of PD and to collect pivotal pre-clinical data demonstrating the safety and efficacy of this novel therapy. This was done in collaboration with our industrial partner (Medos International Sàrl, Le Locle, Switzerland) and with Prof. Stefan Jakob’s group (Bern Insel University Hospital, Switzerland).
In preclinical tests, we are working on the identification of the optimal illumination conditions (wavelength, light dose, light intensity, dose repetition, and delivery route). This is followed by the development of the technology to deliver infrared light to the substantia nigra pars compacta (SNpc) in a safe and controlled way.
We have already built a complete model of light distribution in the human neuronal tissues. This model shows that light can be delivered to the SNpc at an appropriate dose. In parallel, we are investigating the mechanisms of action of PBM in vitro and in vivo in collaboration with our academic partners (Prof. Hilal Lashuel, EPFL, Switzerland and Dr. Patrik Verstreken, VIB, Belgium)
The successful completion of this project will pave the way for the initiation of clinical studies demonstrating the safety and efficacy of the therapy.
Photodynamic drug delivery
Many solid tumors are known to have an abnormal vasculature, which limits the distribution of chemotherapeutic agents. Depending on treatment conditions (irradiance, drug/light doses, the timing between Photosensitizer (PS) administration and irradiation, properties of the PS, etc), photodynamic therapy (PDT) can induce different tissue effects. In most cases, PDT occludes and destroys pathological vasculature.
In our project, conducted in collaboration with the Division of thoracic and vascular surgery, Centre Hospitalier Universitaire Vaudois, Lausanne, Switzerland, we focus on the vascular effects of PDT administered at sub-therapeutic conditions. Thus, we preserve the perfusion of the targeted blood vessels but locally increase their permeability. This leakage is potentially useful to improve the drug delivery of chemotherapeutic agents (such as liposomal doxorubicin (LiporubicinTM)) to tumors. We study this so-called “Photodynamic drug delivery” at the preclinical and clinical level. Our aim is to clarify the different mechanisms involved in PDT-induced leakage; find parameters for optimal and well-controlled drug delivery; bring this novel approach from bench to bedside.
Our group at EPFL is mainly involved in the development of optical fiber-based instruments to deliver and monitor the light inducing this photodynamic drug delivery.
Treatment of atherosclerotic plaques by topical photodynamic therapy
Acute coronary syndromes (ACS) are life-threatening conditions of utmost clinical importance. ACS result from a rupture or erosion of one vulnerable atherosclerotic plaque with secondary platelet activation and thrombus formation, which leads to partial or complete luminal obstruction of the coronary artery. During the last decade, it has been consistently demonstrated that the vascular wall was a very active medium with healing ability. Based on this evidence, it has been suggested that therapies stimulating healing and carried out at the time of percutaneous coronary intervention for ACS could help stabilizing atherosclerotic plaque at risk and decrease the risk of ACS recurrence. Accordingly, photodynamic therapy (PDT) is an interesting concept to be translated into clinical cardiology to trigger appropriate vascular healing. Consequently, the aim of this project conducted in collaboration with the Department of Medicine of the Fribourg University, Fribourg, Switzerland is to select all the necessary tools for the development of vascular PDT by combining specific knowledge of the various research groups involved: in vivo and ex vivo vascular pathophysiology and imaging, photosensitizers, catheters for topical drug and light delivery.