Self-assembly of metal oxalates
The different stages of the particle construction of cobalt oxalate dihydrate from nanoscale to mesoscale were followed using several electron microscopy techniques. The use of cryogenic electron microscopy is a powerful tool in helping us to understand the time dependence aspects of particle growth. Cobalt oxalate dihydrate precipitates in the form of agglomerated rods between 5 and 40 µm in length with an acicular crystalline habit and an organized layered sub-structure.
 |
 |
Firstly, nucleation and growth occur from the supersaturated solution leading to 5-7 nm size partially crystalline primary particles. Then, these primary particles aggregate to form disordered secondary particles. The primary and the secondary particles agglomerate to form the anisotropic core of the final particle. The core acts as template for the third step of growth which corresponds to the self-assembling and alignment, layer by layer around the core, of nanosized crystalline primary particles (~10 nm). The final cobalt oxalate dihydrate particle has a core/shell structure with a partially crystalline core and a self-assembled polycrystalline shell.
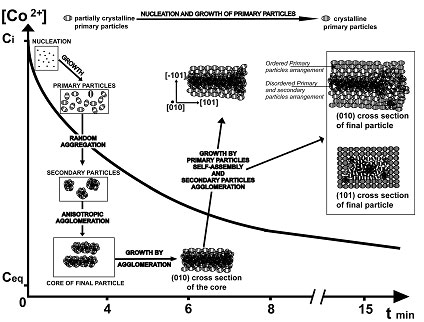
The self-assembly occurs in the last step of growth where we have a lower ionic strength and supersaturation inducing slower kinetics of growth and aggregation. Moreover, the crystalline order in the primary and secondary particles increases with the time of reaction consistent with a continuous process of primary particle nucleation and growth. Population balance modelling should add further weight to these mechanistic propositions.
More information: