M. J. Pougin, N. P. Domingues, F. P. Uran, A. Ortega-Guerrero, C. P. Ireland, J. Espín, W. Lee Queen, and B. Smit, Adsorption in Pyrene-Based Metal–Organic Frameworks: The Role of Pore Structure and Topology ACS Appl. Mater. Interfaces (2024) doi: 10.1021/acsami.4c05527
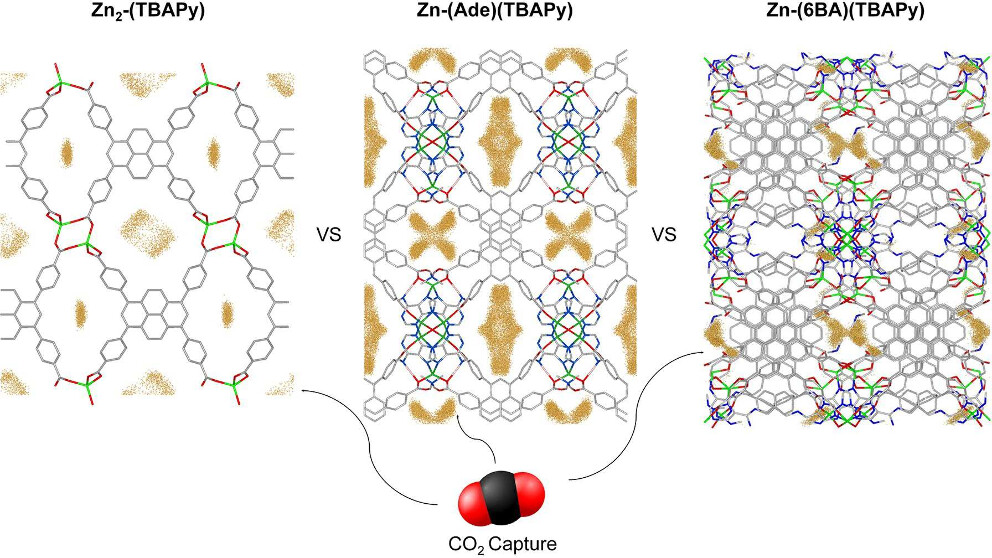
Abstact: Pore topology and chemistry play crucial roles in the adsorption characteristics of metal–organic frameworks (MOFs). To deepen our understanding of the interactions between MOFs and CO2 during this process, we systematically investigate the adsorption properties of a group of pyrene-based MOFs. These MOFs feature Zn(II) as the metal ion and employ a pyrene-based ligand, specifically 1,3,6,8-tetrakis(p-benzoic acid)pyrene (TBAPy). Including different additional ligands leads to frameworks with distinctive structural and chemical features. By comparing these structures, we could isolate the role that pore size, the presence of open-metal sites (OMS), metal–oxygen bridges, and framework charges play in the CO2 adsorption of these MOFs. Frameworks with constricted pore structures display a phenomenon known as the confinement effect, fostering stronger MOF–CO2 interactions and higher uptakes at low pressures. In contrast, entropic effects dominate at elevated pressures, and the MOF’s pore volume becomes the driving factor. Through analysis of the CO2 uptakes of the benchmark materials ─some with narrower pores and others with larger pore volumes─it becomes evident that structures with narrower pores and high binding energies excel at low pressures. In contrast, those with larger volumes perform better at elevated pressures. Moreover, this research highlights that open-metal sites and inherent charges within the frameworks of ionic MOFs stand out as CO2-philic characteristics.